- Home
- Products
- Services
- Product concept development
- Engineering
- Design for manufacturability
- Regulatory services
Read more - Material selection and formulation
- Prototyping
- Testing and validation
Read more - Production process development
- Custom tooling
- Manufacturing
- Finishing operations
- Assembly
- Packaging
- Private labeling
- Sterilization
Read more
- Resources
- Careers
- Search
What is the Eto Sterilization Process and Its Benefits for Exporters?
The Eto Sterilization Process plays a crucial role in the global export industry. This method effectively eliminates microorganisms from products, ensuring their safety and extending shelf life. According to Dr. Jane Thompson, an expert in sterilization technology, "The Eto Sterilization Process is essential for maintaining product integrity in various sectors.”
Many exporters, particularly in the healthcare and food industries, rely on this process. It helps to protect products from contamination during transport. However, not all businesses fully understand the intricacies of this method. The effectiveness of the Eto Sterilization Process can vary, depending on several factors, including temperature and exposure time.
While the advantages are clear, there are challenges to consider. Some products may require specific treatments before sterilization. This adds complexity and may lead to increased costs. Exporters must reflect on these details to ensure they leverage the full potential of the Eto Sterilization Process. Embracing this technology can enhance product safety and marketability, but awareness and preparation are key.

What is Eto Sterilization and How Does It Work?

Ethylene oxide (ETO) sterilization is a process widely used for medical devices and pharmaceuticals. This method employs ethylene oxide gas to eliminate bacteria, viruses, and fungi without damaging sensitive materials. The process occurs within a controlled chamber where items are exposed to ETO gas under specific temperature and humidity conditions.
One of the key benefits of ETO sterilization is its efficacy in ensuring sterility for complex products. It penetrates packaging materials and reaches intricate device designs. Such thorough sterilization is vital in settings where infection control is paramount. However, the process is not without challenges. The gas is toxic and requires careful handling, which can be a logistical hurdle for some manufacturers.
Another critical aspect is the time required for sterilization and aeration. This can delay product availability, posing issues for exporters under tight deadlines. Additionally, concerns around environmental impact and regulatory compliance can complicate the ETO sterilization landscape. As exporters consider ETO, weighing these pros and cons is essential to ensure safe and compliant practices.
Key Applications of Eto Sterilization in Export Industries
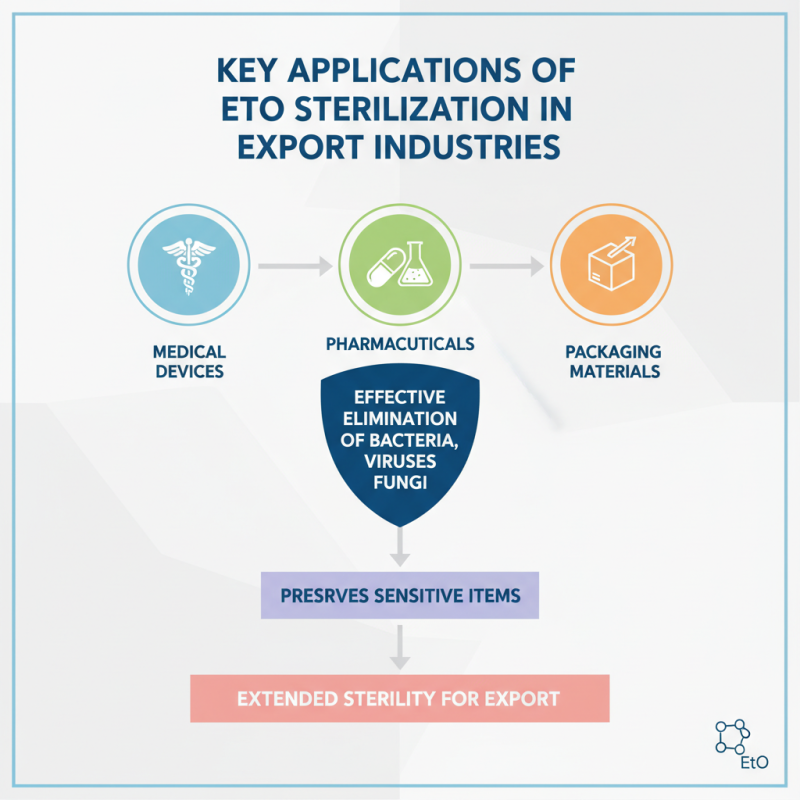
Eto (ethylene oxide) sterilization plays a crucial role in various export industries. It is widely used for medical devices, pharmaceuticals, and packaging materials. This method effectively eliminates bacteria, viruses, and fungi. Its efficiency allows for thorough sterilization without damaging sensitive items. Products treated by Eto remain sterile for extended periods, making them ideal for export markets.
In the food industry, Eto sterilization protects packaged foods from contamination. It extends shelf life and ensures safety. This is particularly important for international shipments, where products face longer transit times. The technique is also used in the textile industry for sterilizing surgical gowns and other fabrics. However, there are challenges. Eto is toxic and requires careful handling, raising safety concerns for workers.
Eto sterilization is not without its imperfections. The necessity for regulatory compliance can be daunting for exporters. Companies must navigate complex guidelines and keep up with evolving regulations. There are instances where improper processing might occur, leading to ineffective sterilization. Continuous monitoring and validation of the process are essential. Nonetheless, the advantages of Eto sterilization make it a valuable option for ensuring product safety in global trade.
Benefits of Utilizing Eto Sterilization for Product Safety
Eto sterilization is a vital method for ensuring product safety, particularly in the export industry. This process involves exposing products to ethylene oxide gas, effectively eliminating harmful microorganisms. By using Eto sterilization, exporters can guarantee that their products are safe for consumers worldwide. This is particularly important as global regulations tighten regarding product safety standards.
Utilizing Eto sterilization offers numerous benefits for exporters. It provides consistent and effective sterilization, safeguarding both products and consumer health. This process is also effective for a wide range of materials, including plastics, textiles, and even electronic devices. The capability to sterilize complex shapes and sizes is a significant advantage, ensuring comprehensive coverage that other methods may miss.
Tips: Always ensure thorough compliance with local safety standards and regulations before choosing this method. Regularly validating the sterilization process can enhance reliability. Keep in mind, however, that the Eto process can take longer than other options, necessitating planning in your production schedules. Balancing efficiency with safety is crucial for maintaining product quality.
Understanding Regulatory Compliance for Eto Sterilization
The EtO sterilization process is crucial for exporters. This method uses ethylene oxide to eliminate microorganisms. It is particularly effective for heat-sensitive products like medical devices. Compliance with regulatory standards is vital in this process. Exporters must understand these regulations to ensure safety and efficacy. Each country has specific rules governing sterilization practices.
Understanding regulatory compliance requires attention to detail. Exporters must document their sterilization process thoroughly. Records should verify that products were treated per the specified protocols. Additionally, regular audits are necessary to maintain compliance. Missteps can lead to significant delays in shipments. Exporters must stay informed about changes in regulations to adapt promptly.
Challenges exist in meeting these compliance standards. Some exporters find it difficult to navigate varying country-specific regulations. This can result in confusion and errors. It is essential to invest in training for staff involved in the compliance process. Awareness of regulatory changes is also key. Ultimately, success in the global market relies on understanding and adhering to these standards.
What is the Eto Sterilization Process and Its Benefits for Exporters? - Understanding Regulatory Compliance for Eto Sterilization
| Dimension | Details |
|---|---|
| Eto Sterilization Method | Ethylene Oxide gas is used to sterilize medical devices and other products. |
| Process Duration | Typically ranges from 6 to 24 hours, depending on load size and type of materials. |
| Temperature | Usually performed at temperatures between 30°C to 60°C. |
| Benefits for Exporters | Ensures compliance with international regulations, increases product shelf life, and enhances marketability. |
| Regulatory Compliance | Meets FDA, ISO, and CE marking requirements for sterility assurance. |
| Environmental Impact | Ethylene Oxide is regulated; proper emissions control is required to minimize environmental risks. |
| Alternatives | Other methods include gamma radiation, electron beam, and steam sterilization. |
Cost-Effectiveness of Eto Sterilization for Exporters
Eto sterilization offers significant cost advantages for exporters looking to maintain product integrity. This process uses ethylene oxide, an effective agent for sterilizing goods without damaging them. According to industry reports, the global demand for ethylene oxide sterilization is projected to reach nearly $600 million by 2026. This indicates a growing recognition of its value in the export sector.
Many manufacturers are drawn to the efficiency of Eto sterilization. It can treat complex medical devices and packaging materials, ensuring compliance with strict health regulations. For exporters, this means reduced logistical costs related to return rates from regulatory rejections. Additionally, studies suggest that Eto treatments can lower the risk of contamination while preserving product functionality, further enhancing the product's market viability.
Though there are clear advantages, exporters must consider the environmental impact. The process requires careful handling and compliance with safety standards, which can generate higher initial costs. Monitoring and managing emissions is vital for sustainability. Balancing cost-effectiveness and environmental responsibility is an ongoing challenge for many in the industry. While Eto sterilization is efficient, exporters need to reflect on its long-term implications for both profitability and ecological health.
Related Posts
-

How to Optimize Your Ethylene Oxide Sterilization Process for Maximum Efficiency
-

Understanding Various Types of Eto Sterilization Methods and Their Ideal Applications
-

Exploring Innovative Alternatives to Best Ethylene Oxide Sterilization Methods
-

Exploring Best Medical Sutures in Surgical Practices and Their Unique Applications
-

Ultimate Guide to Choosing the Best Fep Heat Shrink Tubing for Your Needs
-

Exploring Innovative Autoclave Sterilization Methods for Enhanced Safety and Efficiency






